Mechanical testing of sports shoes
Which mechanical tests on sports shoes does our laboratory offer?
Which types of sports shoes are tested?

In 5 steps to a test report for sports shoes
Why should you have your sports shoes tested by Innoproof?
State-of-the-art testing technology
Our testing laboratory is equipped with state-of-the-art machines, such as the Instron Electropuls E3000. This technology enables precise, reproducible, and dynamic testing at the highest level. This ensures that your products are tested under realistic and precisely controlled conditions.
Owner-managed company with direct communication
As an owner-managed medium-sized company, we offer short decision-making paths and direct communication. Our customers benefit from quick availability and dedicated contacts throughout the entire project. This ensures efficiency, trust, and a collaborative partnership.
Meaningful test reports with added value
Our test reports are not only technically accurate but also clearly structured and easy to understand. High-quality images, clear graphics, and detailed explanations make results transparent and easy to compare. This provides you with a solid basis for decisions in development, quality assurance, and communication.
Sustainable testing without hydraulic oil systems
We deliberately avoid environmentally harmful hydraulic oil systems and rely exclusively on electrodynamic testing systems. This approach is more energy-efficient, cleaner, and requires less maintenance. In this way, we combine state-of-the-art testing technology with responsible use of resources.



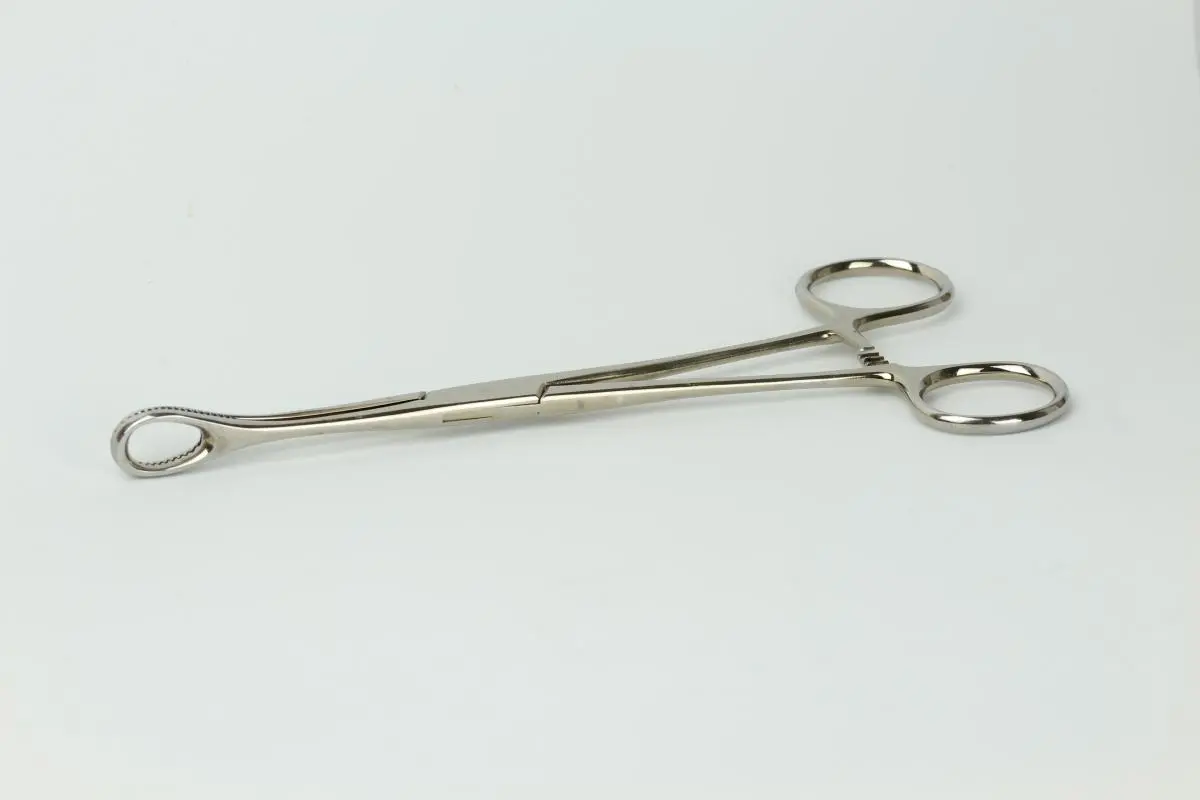
 ASTM F1089 describes physicochemical investigations of surgical instruments with a focus on corrosion.
ASTM F1089 describes physicochemical investigations of surgical instruments with a focus on corrosion.
 The static testing of coating shear strength is described in ASTM F1044. The joining of the specimens and their alignment in the testing machine correspond to those used in dynamic tests.
The opposing, bonded layers are vertically aligned with their contact surface in the loading axis of the testing machine in order to ensure pure shear loading without bending. The specimen holders are mounted on double Cardan joints to decouple constraint forces.
The static testing of coating shear strength is described in ASTM F1044. The joining of the specimens and their alignment in the testing machine correspond to those used in dynamic tests.
The opposing, bonded layers are vertically aligned with their contact surface in the loading axis of the testing machine in order to ensure pure shear loading without bending. The specimen holders are mounted on double Cardan joints to decouple constraint forces. ASTM F1147 specifies a static test setup for determining the tensile or adhesive strength of coatings. The coating is applied to the end face of a cylinder, which is bonded to a second cylinder according to a defined protocol prior to testing.
The opposing bonded layers are axially aligned with their contact surface in the loading axis of the testing machine in order to ensure pure tensile loading. The specimens are mounted on double Cardan joints to decouple constraint forces.
ASTM F1147 specifies a static test setup for determining the tensile or adhesive strength of coatings. The coating is applied to the end face of a cylinder, which is bonded to a second cylinder according to a defined protocol prior to testing.
The opposing bonded layers are axially aligned with their contact surface in the loading axis of the testing machine in order to ensure pure tensile loading. The specimens are mounted on double Cardan joints to decouple constraint forces. We offer measurement of the abrasion resistance of metallic thermal spray coatings using a Taber Abraser in accordance with ASTM F1978-18. The coating under investigation is subjected to controlled pressure and abrasion conditions on rotating discs. The specimen, mounted on a rotating platform, turns about a vertical axis against the sliding rotation of two abrasive wheels. One wheel abrades the specimen outward toward the periphery, while the other abrades inward toward the center.
We offer measurement of the abrasion resistance of metallic thermal spray coatings using a Taber Abraser in accordance with ASTM F1978-18. The coating under investigation is subjected to controlled pressure and abrasion conditions on rotating discs. The specimen, mounted on a rotating platform, turns about a vertical axis against the sliding rotation of two abrasive wheels. One wheel abrades the specimen outward toward the periphery, while the other abrades inward toward the center.
 According to DIN EN 455-1, a water leak test can be performed to verify the watertightness of disposable gloves. Using a vertical tube, 1000 ml of water at a temperature between 15 °C and 35 °C is filled into the glove to be tested. The glove is immediately visually inspected for leaks. The glove is considered non-watertight as soon as water leakage occurs. After 2 to 3 minutes, the glove is visually inspected again.
According to DIN EN 455-1, a water leak test can be performed to verify the watertightness of disposable gloves. Using a vertical tube, 1000 ml of water at a temperature between 15 °C and 35 °C is filled into the glove to be tested. The glove is immediately visually inspected for leaks. The glove is considered non-watertight as soon as water leakage occurs. After 2 to 3 minutes, the glove is visually inspected again. According to DIN EN 455-2, the dimensions and the tensile strength of a medical disposable glove are determined. At least 13 samples per glove batch are used. For dimensional verification, the glove length and width are measured using a ruler and compared with the size table specified in the standard.
According to DIN EN 455-2, the dimensions and the tensile strength of a medical disposable glove are determined. At least 13 samples per glove batch are used. For dimensional verification, the glove length and width are measured using a ruler and compared with the size table specified in the standard.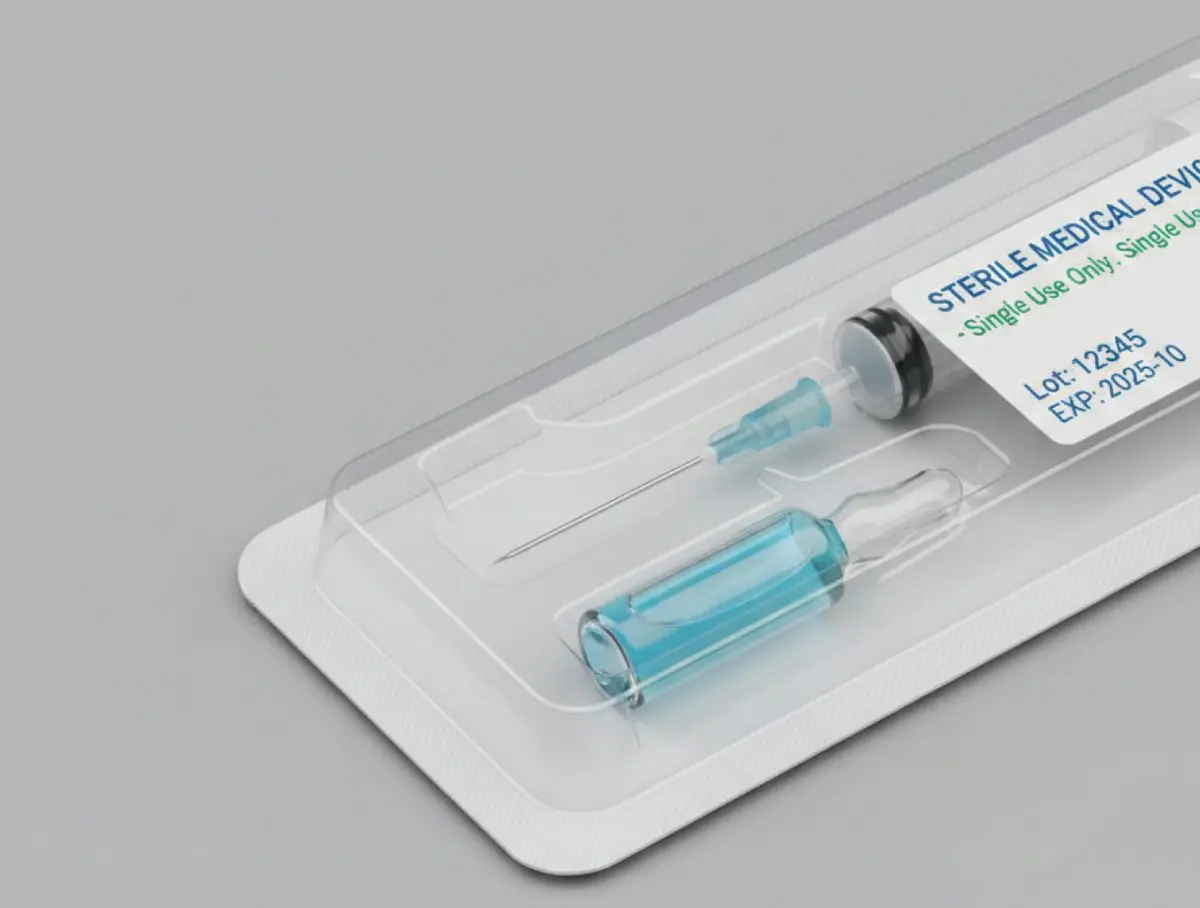
 Packaging is tested under compression according to ASTM D642 in order to simulate transportation or storage conditions. The packaging sample is placed between two horizontal plates and subjected to static loading. The test is terminated when a specified deformation is reached or when a drop in reaction force occurs. The maximum applied test force and the corresponding deformation of the packaging are recorded.
Packaging is tested under compression according to ASTM D642 in order to simulate transportation or storage conditions. The packaging sample is placed between two horizontal plates and subjected to static loading. The test is terminated when a specified deformation is reached or when a drop in reaction force occurs. The maximum applied test force and the corresponding deformation of the packaging are recorded. The seal strength of the packaging seam is tested in a tensile test. The packaging is cut into samples of defined size. Both ends of the seal are clamped, with one of three clamping configurations selected depending on the type of seal. The specimen is statically loaded at 200 to 300 mm/min until seal failure occurs. Only results obtained using the same clamping configuration may be compared.
The seal strength of the packaging seam is tested in a tensile test. The packaging is cut into samples of defined size. Both ends of the seal are clamped, with one of three clamping configurations selected depending on the type of seal. The specimen is statically loaded at 200 to 300 mm/min until seal failure occurs. Only results obtained using the same clamping configuration may be compared.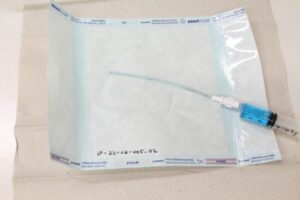 To evaluate the seal seam, dye is applied to one side of the seal. It is then visually examined whether the dye penetrates through the seal. Each side of the sample seal is observed for 5 seconds. The package must not contain liquids or condensation, as this could distort the test results. The dye used must provide strong contrast with the opaque packaging material. One of three methods is selected for seal testing.
To evaluate the seal seam, dye is applied to one side of the seal. It is then visually examined whether the dye penetrates through the seal. Each side of the sample seal is observed for 5 seconds. The package must not contain liquids or condensation, as this could distort the test results. The dye used must provide strong contrast with the opaque packaging material. One of three methods is selected for seal testing. In the bubble emission test according to ASTM F2096, the integrity of packaging or sterile barrier systems is evaluated by applying internal pressure. For this purpose, the packaging film is punctured on one side using a needle, internal air pressure is applied, and the package is submerged in water. In our setup, a second needle and a digital manometer are used to directly measure the internal pressure.
In the bubble emission test according to ASTM F2096, the integrity of packaging or sterile barrier systems is evaluated by applying internal pressure. For this purpose, the packaging film is punctured on one side using a needle, internal air pressure is applied, and the package is submerged in water. In our setup, a second needle and a digital manometer are used to directly measure the internal pressure.
 ISO 5833 specifies, among other things, compression and bending tests for bone cement specimens. We offer static compressive strength testing and static bending strength testing as testing services. For bending strength determination, a four-point bending test is performed. If required, bone cement specimens can be manufactured in our laboratory using our own standard-compliant casting molds.
ISO 5833 specifies, among other things, compression and bending tests for bone cement specimens. We offer static compressive strength testing and static bending strength testing as testing services. For bending strength determination, a four-point bending test is performed. If required, bone cement specimens can be manufactured in our laboratory using our own standard-compliant casting molds.
 We offer various test methods for the mechanical characterization of spinal implants in a vertebrectomy model in accordance with ASTM F1717. The vertebrectomy model simulates the bridging of a vertebral body without anterior support. The spinal implants are rigidly connected to two UHMWPE blocks with well-defined material properties, while a defined gap between the blocks simulates the absence of a vertebral body. The shape and properties of the test blocks are adapted to different regions of the spine (e.g., lumbar or cervical). Together with you, we select the appropriate test procedures for your individual spinal implants from the following options:
We offer various test methods for the mechanical characterization of spinal implants in a vertebrectomy model in accordance with ASTM F1717. The vertebrectomy model simulates the bridging of a vertebral body without anterior support. The spinal implants are rigidly connected to two UHMWPE blocks with well-defined material properties, while a defined gap between the blocks simulates the absence of a vertebral body. The shape and properties of the test blocks are adapted to different regions of the spine (e.g., lumbar or cervical). Together with you, we select the appropriate test procedures for your individual spinal implants from the following options:
 We offer various test methods for the mechanical characterization of spinal fusion implants in accordance with ASTM F2077. Together with you, we select the appropriate test procedures for your individual fusion implants from the following options:
We offer various test methods for the mechanical characterization of spinal fusion implants in accordance with ASTM F2077. Together with you, we select the appropriate test procedures for your individual fusion implants from the following options:

 We offer a testing procedure in accordance with ASTM F382 for comparing bone plates with respect to their mechanical properties. Specifically, the following two tests can be performed
We offer a testing procedure in accordance with ASTM F382 for comparing bone plates with respect to their mechanical properties. Specifically, the following two tests can be performed We offer characterization of angled plates in accordance with ASTM F384. Specifically, we can perform static and dynamic bending tests:
We offer characterization of angled plates in accordance with ASTM F384. Specifically, we can perform static and dynamic bending tests: We offer various test methods for the mechanical characterization and classification of bone screws in accordance with ASTM F543. Together with you, we select the appropriate test procedures from the following range to suit your specific screw design:
We offer various test methods for the mechanical characterization and classification of bone screws in accordance with ASTM F543. Together with you, we select the appropriate test procedures from the following range to suit your specific screw design: We offer characterization of the design and mechanical performance of intramedullary nails in accordance with ASTM F1264.
We offer characterization of the design and mechanical performance of intramedullary nails in accordance with ASTM F1264.
 We offer a testing procedure to evaluate the fixation strength of the glenoid component in bone and its resistance to subluxation caused by cyclic movement (e.g., superior-inferior or anterior-posterior) of the humeral head against the edge of the glenoid, according to the ASTM F2028 standard. This test method can be applied to both cemented monolithic and modular glenoid components as well as uncemented reverse glenoid components.
We offer a testing procedure to evaluate the fixation strength of the glenoid component in bone and its resistance to subluxation caused by cyclic movement (e.g., superior-inferior or anterior-posterior) of the humeral head against the edge of the glenoid, according to the ASTM F2028 standard. This test method can be applied to both cemented monolithic and modular glenoid components as well as uncemented reverse glenoid components.